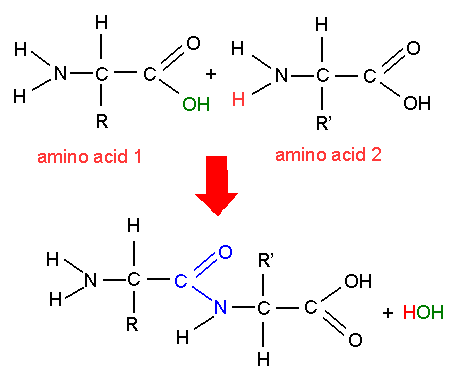
It is also possible for the same elements to combine in the same proportions but in a different physical configuration. As an example, two hydrogen atoms and one oxygen atom form water (H 2O), while two hydrogen atoms and two oxygen atoms form hydrogen peroxide (H 2O 2). The same atoms may combine in different proportions to form different molecules. Larger molecules are called polymers and may be made of thousands of atoms.Ītoms can combine in many different ways as molecules. Polyatomic molecules have more than two atoms, such as water (H 2O) and carbon dioxide (CO 2). 
If the two atoms are the same element, it is called a homonuclear diatomic molecule, such as oxygen (O 2) and nitrogen (N 2). An example of a diatomic molecule is carbon monoxide (CO) made of a single atom of carbon and one of oxygen. If the covalent bond is broken and the molecule is split apart, it requires energy input and is thereby endothermic.ĭiatomic molecules are when only two atoms combine. When atoms bond, energy is released in an exothermic reaction. When two or more atoms come together to share outer shell valence electrons, a chemical (covalent) bond is formed, and they enter a lower energy state. When an atom has an incomplete electron shell, it is said to have valence electrons. They consist of a nucleus and surrounding electrons. Molecules are noted with the element symbol and a subscript with the number of atoms.Ītoms are the fundamental unit of an element. 
A molecule is two or more atoms connected by chemical bonds, which form the smallest unit of a substance that retains the composition and properties of that substance.
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
